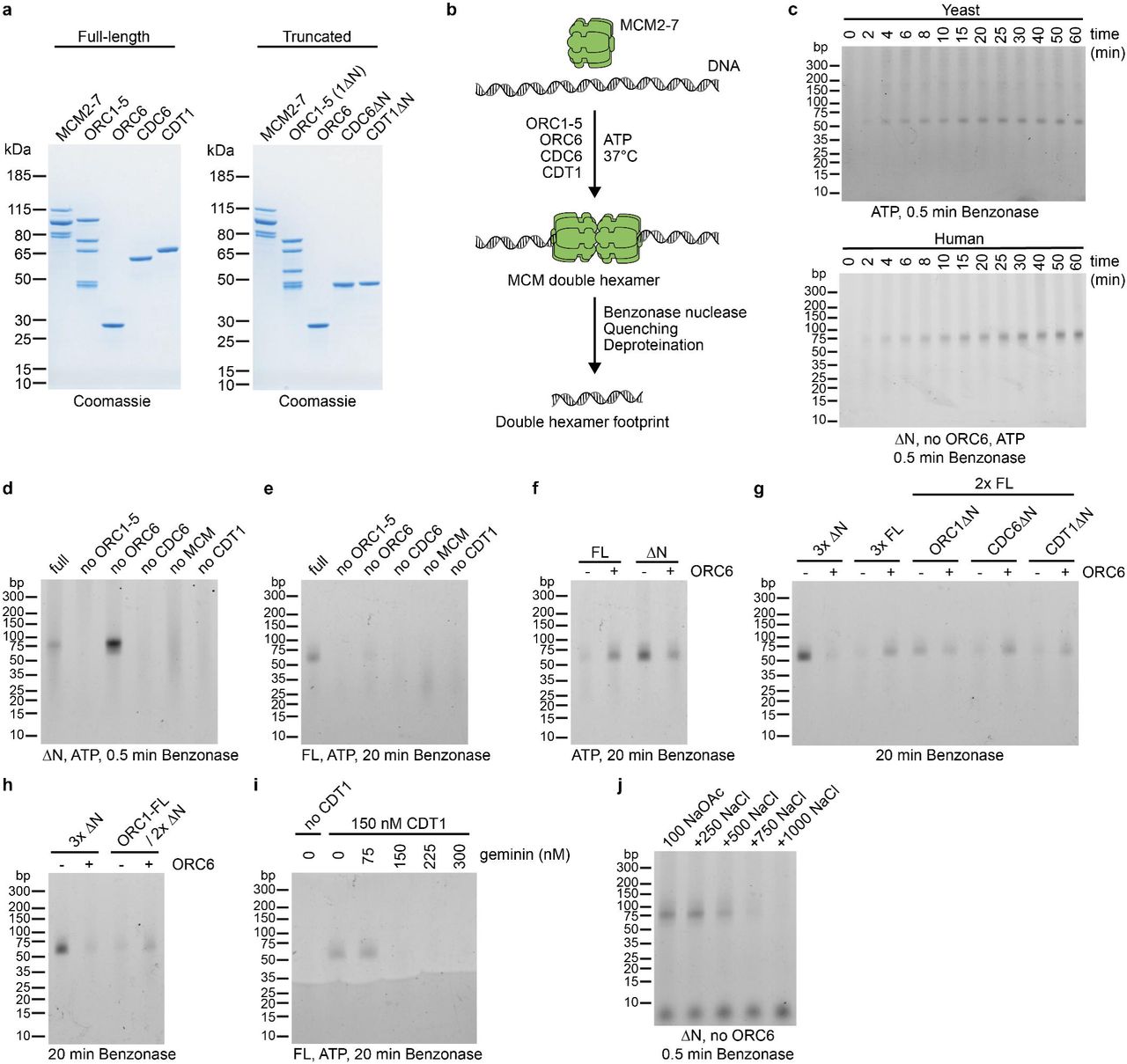
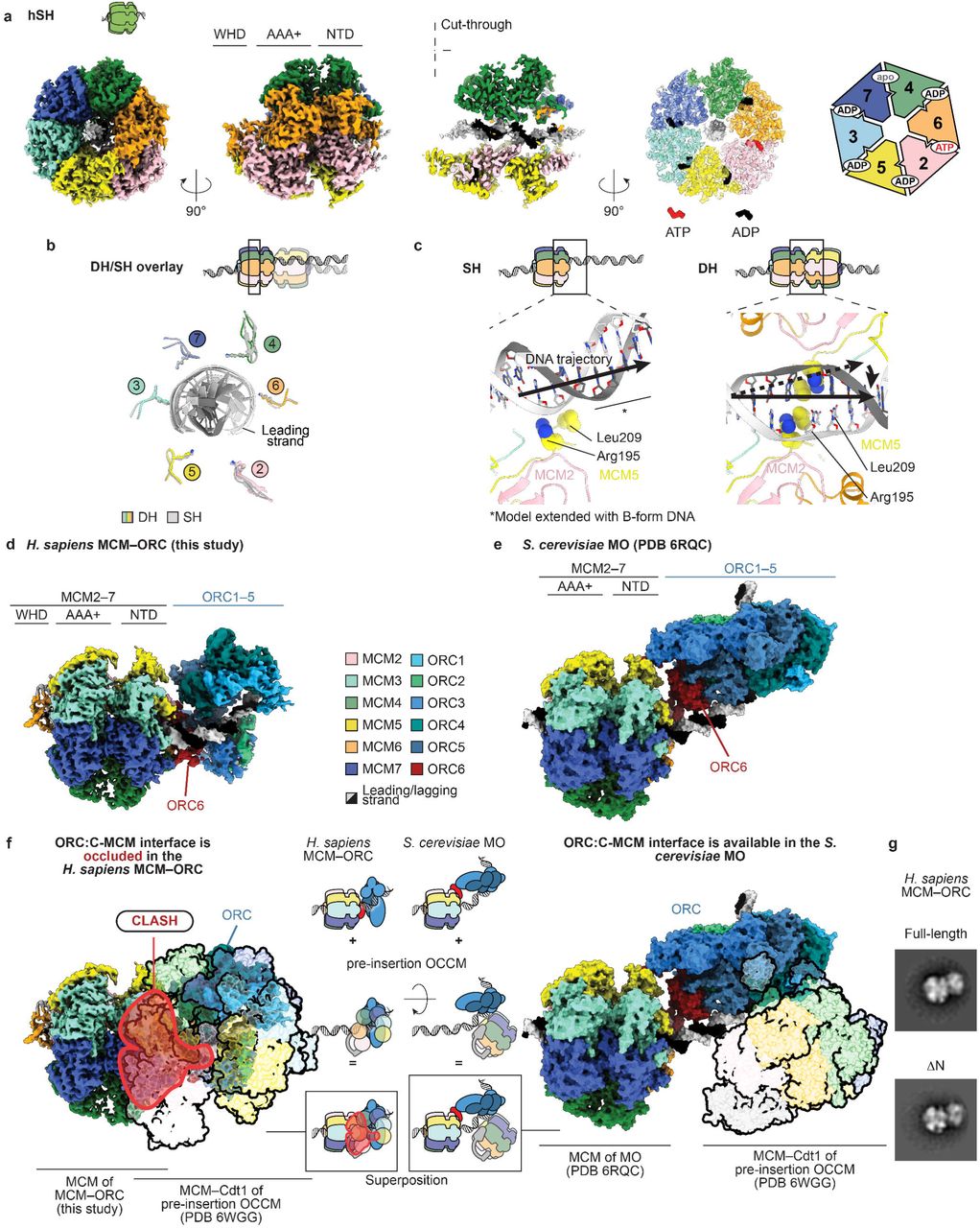
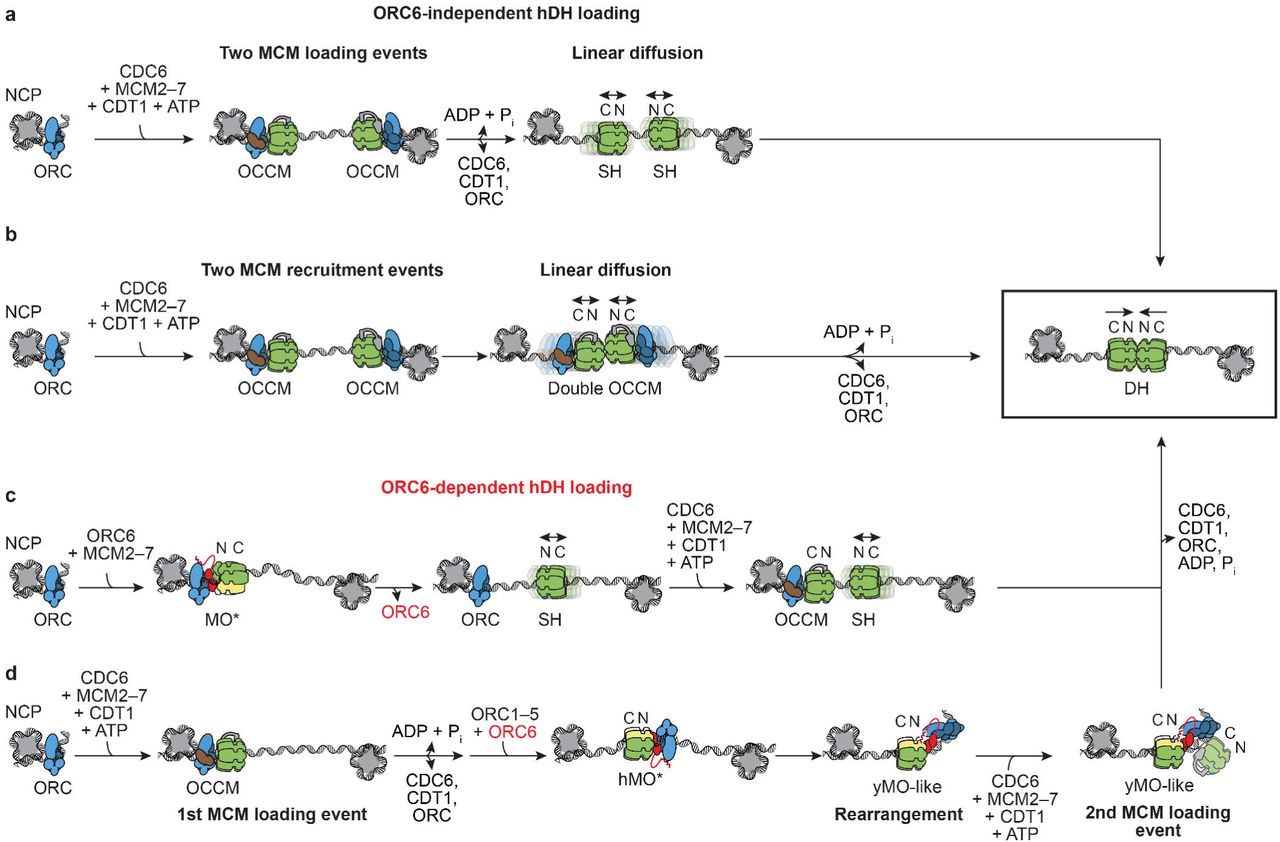
orc5 (Boster Bio)
Structured Review

Orc5, supplied by Boster Bio, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more
https://www.bioz.com/result/orc5/product/Boster Bio
Average 93 stars, based on 1 article reviews
Images
1) Product Images from "DNA replication in primary hepatocytes without the six-subunit ORC"
Article Title: DNA replication in primary hepatocytes without the six-subunit ORC
Journal: eLife
doi: 10.7554/eLife.102915

Figure Legend Snippet: ( A ) Scheme of introduced loxP sites in Orc 2 locus. ( B ) Representative picture of genotyping of offspring coming from Orc2 f/+ crossed with Orc2 f/+ . ( C ) The ratio of observed to expected animals coming from Orc2 f/+ crossed with Orc2 f/+ . ( D ) Schematic of the ORC2 protein and the DeltaORC2 protein produced after deletion of exons 6 and 7. A110 is mutated to V110 and then the protein goes out of frame. ( E ) Validation of Orc2 deletion 3 d after Adeno cre transduction. ( F ) Western blot of ORC2 protein 5 d after Adeno cre transduction. 10 or indicated μl of lysate loaded/lane as written on the top. ( G ) MTT assay of WT and Orc2 f/f MEFs without and with Adeno cre transduction. ( H ) Western blot of ORC2 protein 5 and 15 d after Adeno Cre transduction. Figure 1—source data 1. PDF file containing original DNA gel picture corresponding to , panel B, indicating the relevant bands and individual animals. Figure 1—source data 2. Original image for , panel B. Figure 1—source data 3. PDF file containing original DNA gel picture corresponding to , panel E, indicating the relevant bands and increasing Adeno-Cre. Figure 1—source data 4. Original image for , panel E. Figure 1—source data 5. PDF file containing original Western blot membrane picture corresponding to , panel F, indicating the relevant bands and addition of Adeno-Cre. Figure 1—source data 6. Original image for , panel F. Figure 1—source data 7. PDF file containing original Western blot membrane picture corresponding to , panel H, indicating the relevant bands and ORC2 protein expression. Figure 1—source data 8. Original image for , panel H.
Techniques Used: Produced, Biomarker Discovery, Transduction, Western Blot, MTT Assay, Membrane, Expressing

Figure Legend Snippet: ( A ) Scheme of Alb +/- - Orc2 f/f ROSA26 stop-EYFP crossed with Alb +/- -Orc2 f/f ROSA26 stop-EYFP (All mice are with ROSA26 stop-EYFP and so we do not include this in the genotypes below). ( B ) The ratio of observed to expected animals coming from A. ( C ) Western blot of hepatocytes from Orc2 f/f and Alb +/- - Orc2 f/f animals. Tubulin was used as loading control. ( D ) Quantification of the Western blots of hepatocyte lysates from Orc2 f/f (without Alb-cre ) mice and the same genotype but with Alb-Cre to show the levels of other key replication initiation proteins in the ORC2 KO hepatocytes. ( E ) Average body weight of Orc2 f/f and Alb-Orc2 f/f animals. ( F ) Average liver weight of Orc2 f/f and Alb-Orc2 f/f animals. ( G ) Average liver-to-body weight ratio of Orc2 f/f and Alb-Orc2 f/f animals. ( H ) Representative H&E staining of liver tissue from Orc2 f/f (WT) and Alb-Orc2 f/f (KO) animals. Both panels at same scale. ( I ) Quantification of hepatocyte nuclear size in Orc2 f/f and Alb-Orc2 f/f animals. ( J ) Quantification of hepatocyte nuclear size in Orc2 f/f and Alb-Orc2 f/f female mice. ( K ) Quantification of hepatocytes nuclear size in Orc2 f/f and Alb-Orc2 f/f male mice. *p<0.05, **p<0.01, two-tailed Student’s t-test. Figure 2—source data 1. Original Western blot membrane picture corresponding to , panel C. Molecular weight markers are labeled on the left. The bands next to the arrow represent ORC2 protein. Figure 2—source data 2. Original image for , panel C. Figure 2—source data 3. Original Western blot membrane picture corresponding to , panel C. Molecular weight markers are labeled on the left. The bands next to the arrow represent Tubulin protein. Figure 2—source data 4. Original image for , panel C.
Techniques Used: Western Blot, Control, Staining, Two Tailed Test, Membrane, Molecular Weight, Labeling

Figure Legend Snippet: ( A–B ) Breeding schemes to obtain conditional double flox animals. ( C ) The ratio of observed to expected animals coming from B. Orc1 =all animals with Orc1 f/f ROSA26 stop-EYFP , Orc2 =all animals with Orc2 f/f ROSA26 stop-EYFP , Orc1 Orc2 =all animals with Orc1 f/f Orc2 f/f ROSA26 stop-EYFP genotype. This was before the introduction of Alb-Cre . ( D ) Immunoblot of hepatocytes from WT ( Orc1 f/f Orc2 f/f ) and DKO ( Orc1 f/f Orc2 f/f Alb-cre +/- ) mice to show that ORC1 and ORC2 are depleted in the DKO cells. ( E ) Quantitation of immunoblots to show that levels of other key initiation protein subunits are not decreased in the DKO mice hepatocytes. ( F ) Average body, liver weight, and their ratio for WT and DKO animals. ( G ) Representative H&E staining of liver tissue from male WT and DKO animals. ( H ) Quantification of hepatocyte nuclear size in the WT and DKO animals. ( I ) Quantification of nuclei ploidy for EYFP low (includes negative) and high (positive) primary liver cells from DKO mice. Figure 6—source data 1. PDF file containing original Western blot membrane picture corresponding to , panel D, indicating the relevant bands, ORC1 protein expression, and individual animals. Figure 6—source data 2. Original image for panel D, ORC1 protein expression, and individual animals. Figure 6—source data 3. PDF file containing original Western blot membrane picture corresponding to , panel D, indicating the relevant bands, ORC2 protein expression, and individual animals. Figure 6—source data 4. Original image for panel D, ORC2 protein expression, and individual animals. Figure 6—source data 5. PDF file containing original Western blot membrane picture corresponding to , panel D, indicating the relevant bands, HSP90 protein expression, and individual animals. Figure 6—source data 6. Original image for panel D, HSP90 protein expression, and individual animals.
Techniques Used: Western Blot, Quantitation Assay, Staining, Membrane, Expressing